Thermal Energy and Matter

Thermal energy is the combined amount of potential and kinetic energy of particles in an object. It depends on the temperature, phase, and mass of an object. Substances can have the same temperature, and still be able to have different amounts of thermal energy. A great example of thermal energy is the suns energy.
Work and Heat-
Heat is a transfer of thermal energy between two objects which results in a difference in temperature. It flows constantly from hot to colds items and is produced by friction.
Temperature-
Temperature is the measure of how cool or warm something is compared to a reference point. Temperature can be easily read using a thermometer which can tell temperature using Fareinheit and Celsius scales. Unlike the Fareinheit scale, on the Celsius the reference points for temperature are the boiling and freezing points of water. When an object heats up, the particles of the object begin to move faster, and the temperature increase.
Thermal Expansion and Contraction-
Thermal expansion occurs when matter is heated. This is because the particles move farther apart from eachother as temperature increases. Also in thermal expansion the space between the particles increases as well as the amount of volume. Unlike thermal expansion, thermal contractions temperature decreases, along with its space between particles and its volume.
Specific Heat-
Specific heat is the amount of heat that is needed to increase the temperature of a gram of material by a one degree Celsius. When an object has a lower specific heat, it will have an increase in temperature when the heat is observed.
Measuring Heat Changes-
To measure heat changes in thermal energy you would use a device called a calorimeter. A calorimeter uses the principle that heat flows from a hotter object to a colder one until the objects reach the exact same temperature.
Work and Heat-
Heat is a transfer of thermal energy between two objects which results in a difference in temperature. It flows constantly from hot to colds items and is produced by friction.
Temperature-
Temperature is the measure of how cool or warm something is compared to a reference point. Temperature can be easily read using a thermometer which can tell temperature using Fareinheit and Celsius scales. Unlike the Fareinheit scale, on the Celsius the reference points for temperature are the boiling and freezing points of water. When an object heats up, the particles of the object begin to move faster, and the temperature increase.
Thermal Expansion and Contraction-
Thermal expansion occurs when matter is heated. This is because the particles move farther apart from eachother as temperature increases. Also in thermal expansion the space between the particles increases as well as the amount of volume. Unlike thermal expansion, thermal contractions temperature decreases, along with its space between particles and its volume.
Specific Heat-
Specific heat is the amount of heat that is needed to increase the temperature of a gram of material by a one degree Celsius. When an object has a lower specific heat, it will have an increase in temperature when the heat is observed.
Measuring Heat Changes-
To measure heat changes in thermal energy you would use a device called a calorimeter. A calorimeter uses the principle that heat flows from a hotter object to a colder one until the objects reach the exact same temperature.
Heat and Thermodynamics-
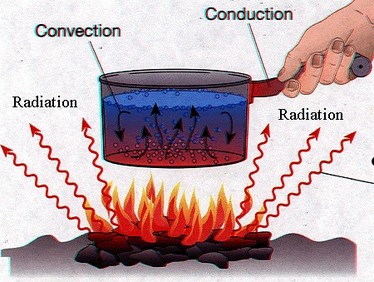
Conduction-
Conduction is a transfer of thermal energy that does not have an overall transfer of matter. Conduction occurs slower when it is occuring in gases instead of liquids or solids. This is because the particles in a gas collide less often. Conduction occurs fastest when in metals than any other solid, because metals have free electrons that can easily transfer thermal energy. When in most solids conduction takes place as particles vibrate in place. Conduction has thermal conductors and thermal insulators. Thermal conductors are able to conduct energy very well, while thermal insulators conduct energy very poorly. Examples of thermal conductors are copper and aluminum. Examples of thermal insulators, are items such as wood, foam cups, and even air. Air is an good example of a thermal insulator because, air can slow down conduction to reduce heat loss in the winter, and can help to keep heat from getting inside somewhere in the summer.
Convection-
Covection is the transfer of thermal energy when particles from a fluid move from place to place. Have you ever wondered why the heat at the bottom of a convection oven is greater then at the top? This is because of the convection currents that form causing the heat to rise and cool as it rises. A convection current is when some type of fluid circulates in a loop causing an object to get hotter or colder. You may not know it, but we often see convection currents in nature, such as ocean currents, weather systems, and when hot rock has movement in our Earth's interior.
Radiation-
Radiation is the transfer of thermal energy by waves that move throughout space. It is possible for all objects to radiate some type of energy through radiation. Some objects can recieve less radiation than others by being farther away from a radiating object. A lot of radiation is received from the suns rays.
Conduction is a transfer of thermal energy that does not have an overall transfer of matter. Conduction occurs slower when it is occuring in gases instead of liquids or solids. This is because the particles in a gas collide less often. Conduction occurs fastest when in metals than any other solid, because metals have free electrons that can easily transfer thermal energy. When in most solids conduction takes place as particles vibrate in place. Conduction has thermal conductors and thermal insulators. Thermal conductors are able to conduct energy very well, while thermal insulators conduct energy very poorly. Examples of thermal conductors are copper and aluminum. Examples of thermal insulators, are items such as wood, foam cups, and even air. Air is an good example of a thermal insulator because, air can slow down conduction to reduce heat loss in the winter, and can help to keep heat from getting inside somewhere in the summer.
Convection-
Covection is the transfer of thermal energy when particles from a fluid move from place to place. Have you ever wondered why the heat at the bottom of a convection oven is greater then at the top? This is because of the convection currents that form causing the heat to rise and cool as it rises. A convection current is when some type of fluid circulates in a loop causing an object to get hotter or colder. You may not know it, but we often see convection currents in nature, such as ocean currents, weather systems, and when hot rock has movement in our Earth's interior.
Radiation-
Radiation is the transfer of thermal energy by waves that move throughout space. It is possible for all objects to radiate some type of energy through radiation. Some objects can recieve less radiation than others by being farther away from a radiating object. A lot of radiation is received from the suns rays.
Thermodynamics-
Thermodynamics is where conversions between thermal enegy and other types of energy is studied. There are three laws of thermodynamics. From thermodynamics we learn that energy can be converted into different forms, but it can't be destroyed or created. This is known as the "First Law of Thermodynamis", which states that energy is something that is conserved. Thermodynamics also simply tells us that thermal energy flows from hotter to colder objects, but only when work is done on the object. When thermal energy is unable to be transformed into work it is known as waste heat. This is the Second Law of Thermodynamics. The "Third Law of Thermodynamics" says that absolute zero can't be reached when cooling matter. Although absolute zero can't be reached scientists have been able to almost reach absolute zero, but not quite.
Thermodynamics is where conversions between thermal enegy and other types of energy is studied. There are three laws of thermodynamics. From thermodynamics we learn that energy can be converted into different forms, but it can't be destroyed or created. This is known as the "First Law of Thermodynamis", which states that energy is something that is conserved. Thermodynamics also simply tells us that thermal energy flows from hotter to colder objects, but only when work is done on the object. When thermal energy is unable to be transformed into work it is known as waste heat. This is the Second Law of Thermodynamics. The "Third Law of Thermodynamics" says that absolute zero can't be reached when cooling matter. Although absolute zero can't be reached scientists have been able to almost reach absolute zero, but not quite.
Quiz-
See what you have learned from the article above, and take the quiz.
1) True/False: Temperature is related to the average amount of kinetic energy in an object.
2) What is thermal expansion?
a. The decrease in volume of a material due to a temperature increase.
b. The increase in volume of a material due to a temperature decrease.
c. The increase in volume of a material due to a temperature increase.
d. The decrease in volume of a material due to a temperature decrease.
3) The amount of heat needed to raise the temperature of one gram of a material by one degree Celsius, describes which of the following?
a. Measuring Heat Changes
b. Work and Heat
c. Thermal Energy
d. Spedific Heat
4) True/False: Conduction is faster in gases, than in solids and liquids.
5) True/False: A thermal conductor is a material that conducts thermal energy very well.
6) True/False: All objects do not radiate energy.
Answers:
1) True
2) C
3) D
4) False
5) True
6) False
1) True/False: Temperature is related to the average amount of kinetic energy in an object.
2) What is thermal expansion?
a. The decrease in volume of a material due to a temperature increase.
b. The increase in volume of a material due to a temperature decrease.
c. The increase in volume of a material due to a temperature increase.
d. The decrease in volume of a material due to a temperature decrease.
3) The amount of heat needed to raise the temperature of one gram of a material by one degree Celsius, describes which of the following?
a. Measuring Heat Changes
b. Work and Heat
c. Thermal Energy
d. Spedific Heat
4) True/False: Conduction is faster in gases, than in solids and liquids.
5) True/False: A thermal conductor is a material that conducts thermal energy very well.
6) True/False: All objects do not radiate energy.
Answers:
1) True
2) C
3) D
4) False
5) True
6) False